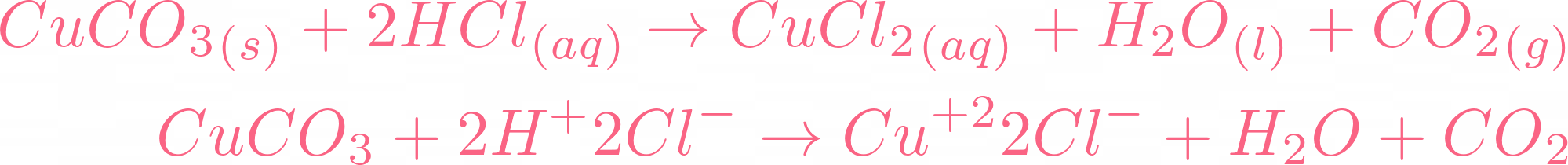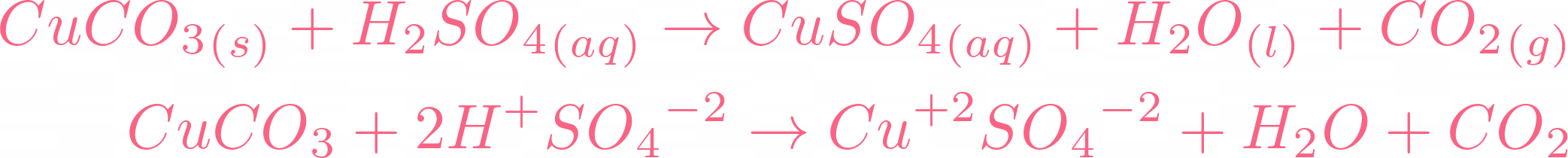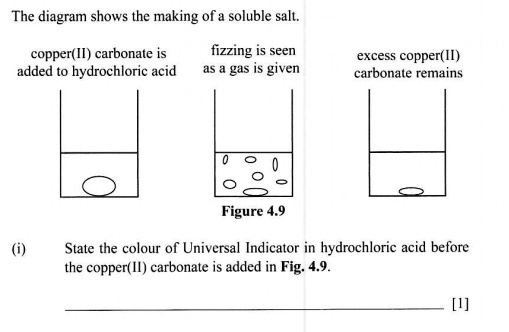
SOLVED: Please ensure that your responses are ACCURATE and DETAILED and please give EXPLANATIONS. The diagram shows the making of a soluble salt: copper(II) carbonate is added to hydrochloric acid fizzing is
7. 0 g of copper (ll) carbonate is added to 50cm3 of 1.0mol dm-3 hydrochloric acid. What is the mass of copper (ll) carbonate left at the end of the reaction? - Quora

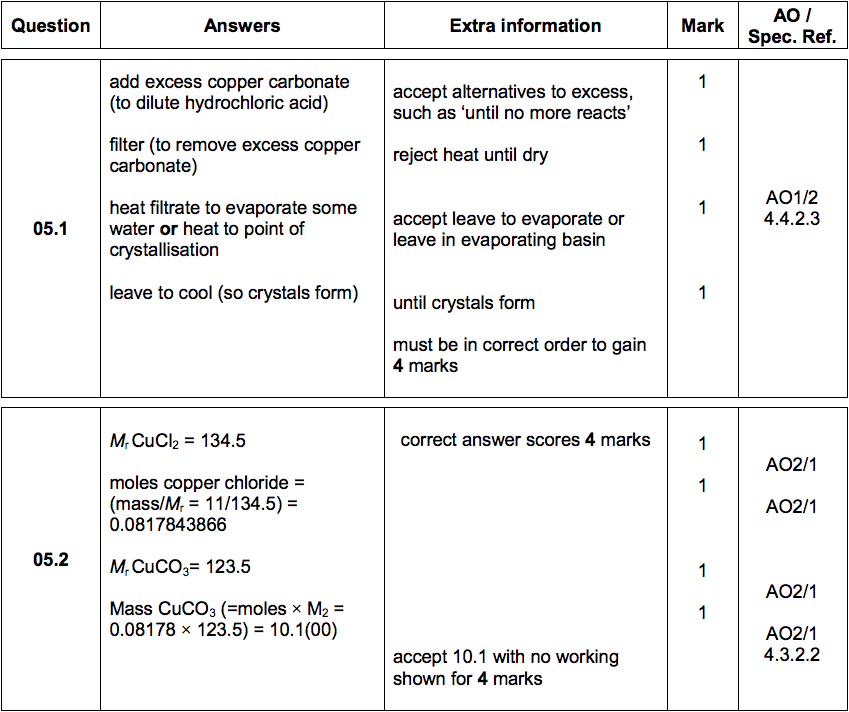
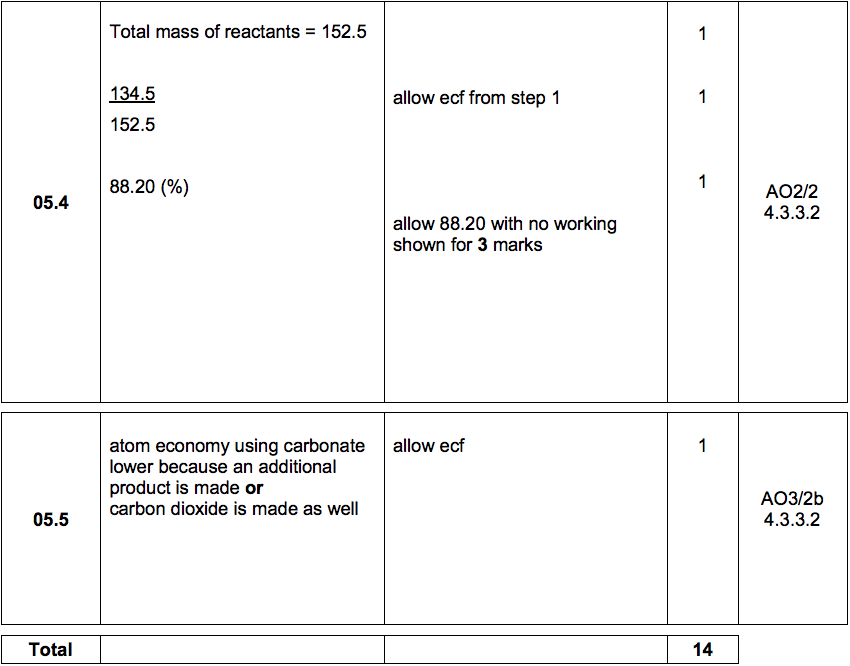
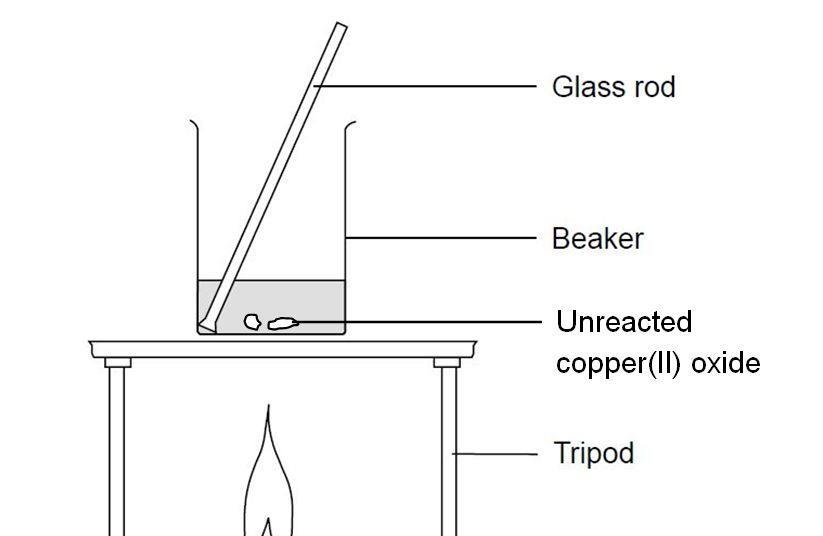
A student investigated the reactions of copper carbonate and copper oxide with dilute hydrochloric acid. How can a sample of copper chloride crystals be made from copper carbonate and dilute hydrochloric acid?
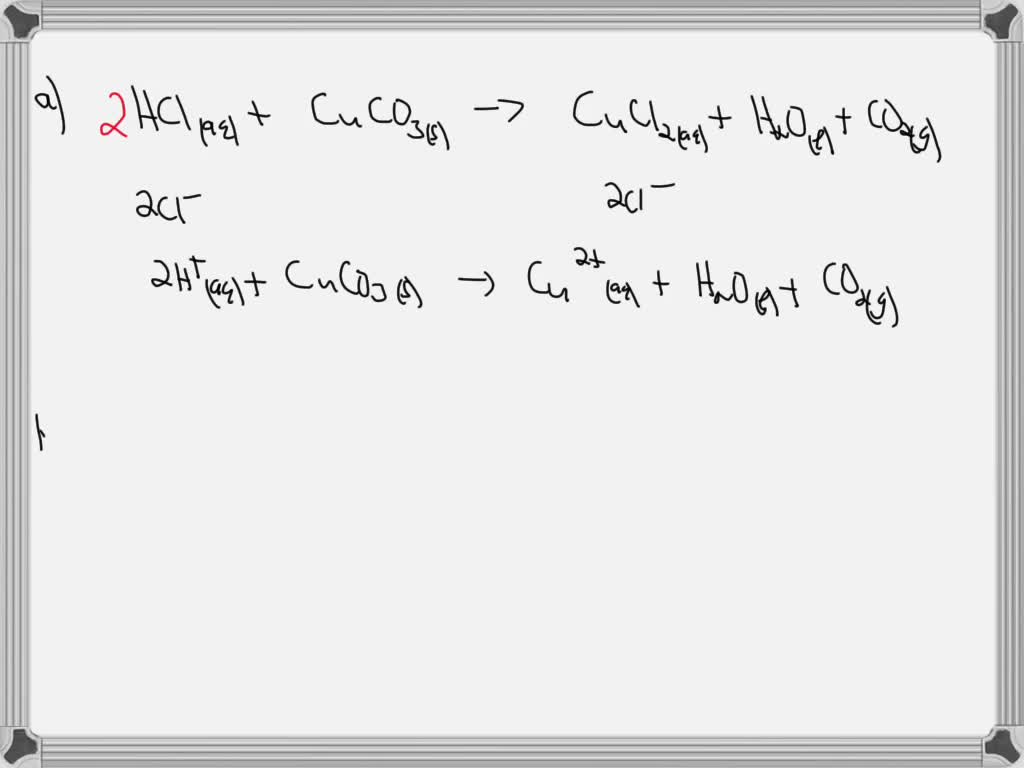
SOLVED: A. Write a net ionic equation for the reaction that occurs when excess hydrochloric acid (aq) and copper(II) carbonate are combined. B. Write a net ionic equation for the reaction that