
Pa, which would be produced when 1.25 g of calcium carbonate reacts completely with the hydrochloric acid.
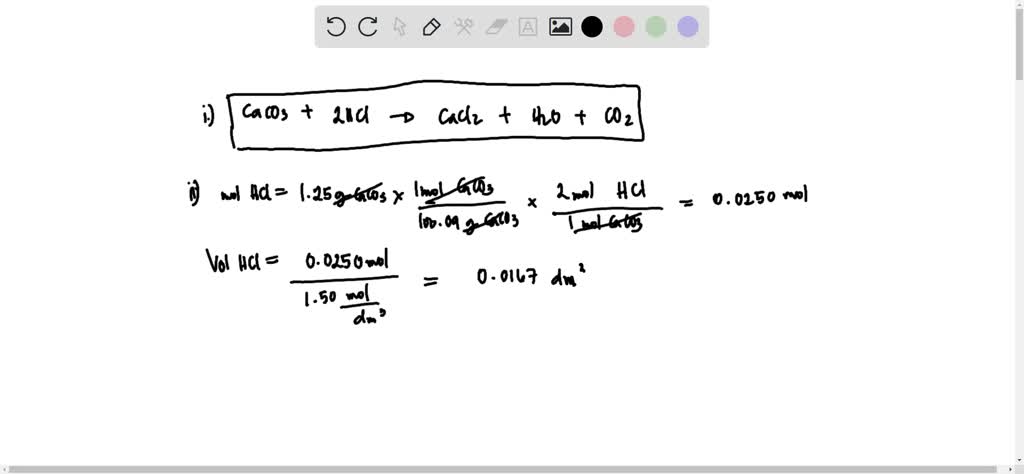
SOLVED: Calcium carbonate is added to separate solutions of hydrochloric acid and ethanoic acid of the same concentration. State one similarity and one difference in the observations you could make. (i) Write

When Ethanoic Acid Reacts with Sodium Hydrogen Carbonate, Then a Salt X is Formed and a Gas Y is Evolved. Name the Salt X and Gas. Y Describe an Activity with the

pKa Ka data factors affecting Acidic reactions of carboxylic acids with metals oxides hydroxides carbonates hydrogencarbonate test advanced A level organic chemistry revision notes doc brown

Writing a Net Ionic Equation for the Reaction of Solid Calcium Carbonate with a Hydrochloric Acid Solution

Reaction 3 - Reaction of ethanoic acid with calcium carbonate - 7. Investigate the reactions of carboxylic acids - GCSE Chemistry (Single Science) Revision - CCEA - BBC Bitesize
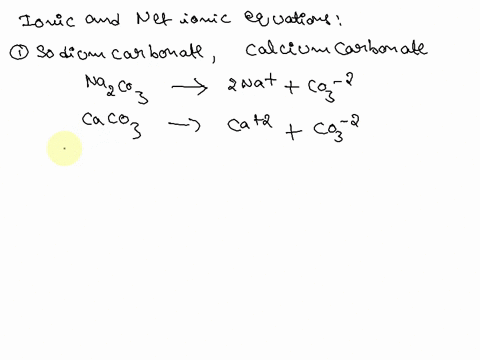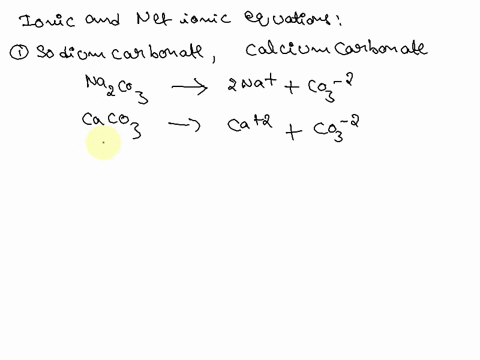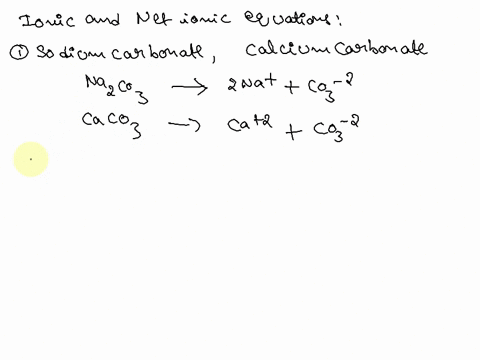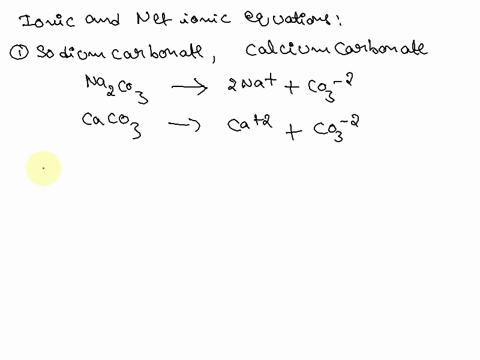

.jpg)



![Carbon] Ethanoic acid - Formation, Properties, Uses [with Reactions] Carbon] Ethanoic acid - Formation, Properties, Uses [with Reactions]](https://d1avenlh0i1xmr.cloudfront.net/fb1b0fbe-d74a-44bb-8d4d-1b41b5aa53a2/reaction-of-ethanoic-acid-with-sodium-carbonate-and-sodium-bicarbonate---teachoo.jpg)



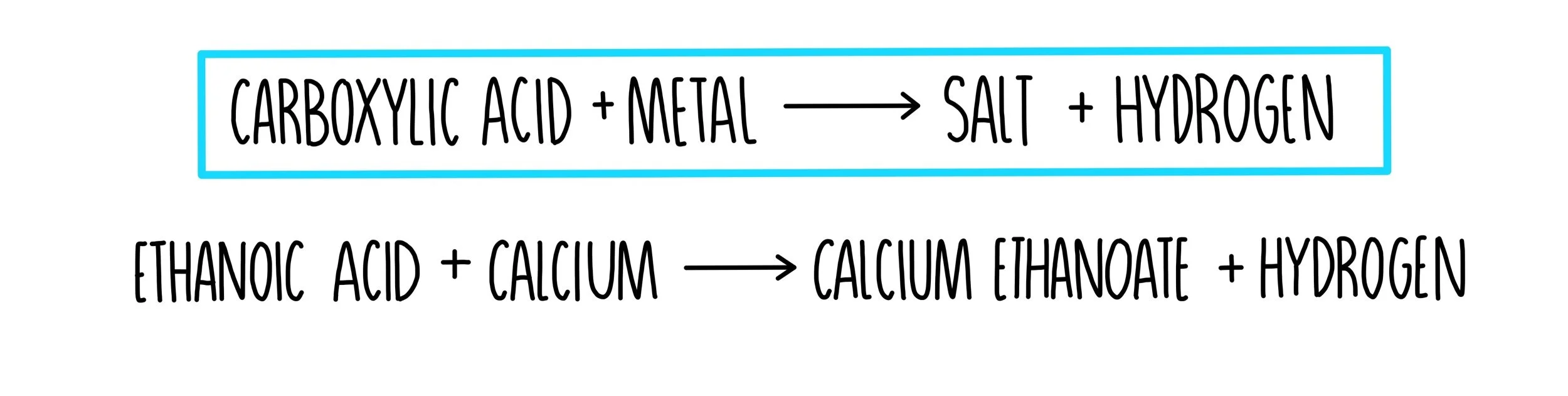


.jpg)

