\\[\\left( {{\\text{N}}{{\\text{H}}_4}} \\right){\\text{S}}{{\\text{O}}_4}\\](C) \\[{\\text{N}}{{\\text{a}}_2}{\\text{C}}{{\\text{O}}_3}\\](D) \\[{\\left( {{\\text{N ... The molecular formula of sodium carbonate is:(A) \\[{\\text{NaC}}{{\\text{O}}_3}\\](B) \\[\\left( {{\\text{N}}{{\\text{H}}_4}} \\right){\\text{S}}{{\\text{O}}_4}\\](C) \\[{\\text{N}}{{\\text{a}}_2}{\\text{C}}{{\\text{O}}_3}\\](D) \\[{\\left( {{\\text{N ...](https://www.vedantu.com/question-sets/2e37659b-297f-488b-bc60-a7c4facc2cff8373828392292757040.png)
The molecular formula of sodium carbonate is:(A) \\[{\\text{NaC}}{{\\text{O}}_3}\\](B) \\[\\left( {{\\text{N}}{{\\text{H}}_4}} \\right){\\text{S}}{{\\text{O}}_4}\\](C) \\[{\\text{N}}{{\\text{a}}_2}{\\text{C}}{{\\text{O}}_3}\\](D) \\[{\\left( {{\\text{N ...

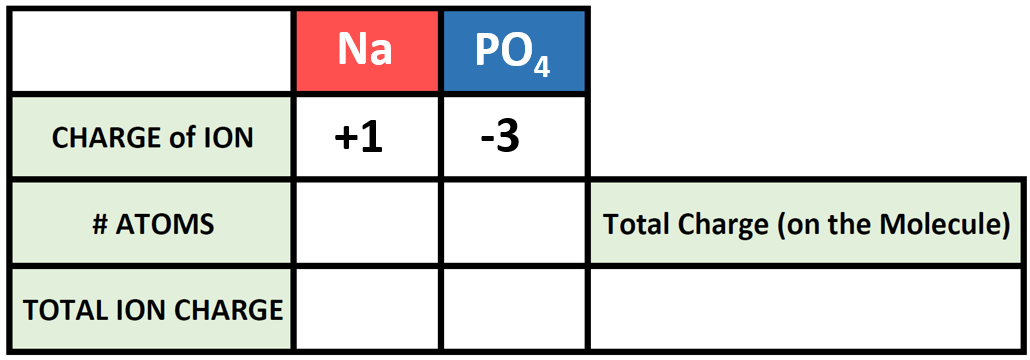
What are polyatomic ions? n Examples: n CO 3 2- = carbonate. n CO 2 2- = carbonite. n PO 4 3- = phosphate n PO 3 3- = phosphite Polyatomic ions are ions. - ppt download
Negatively charged components of oil (a) detached from the positive... | Download Scientific Diagram

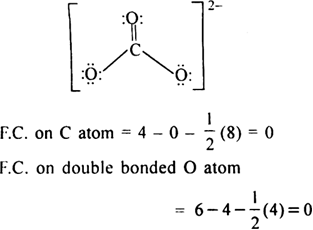
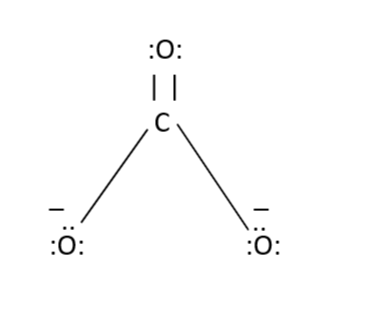
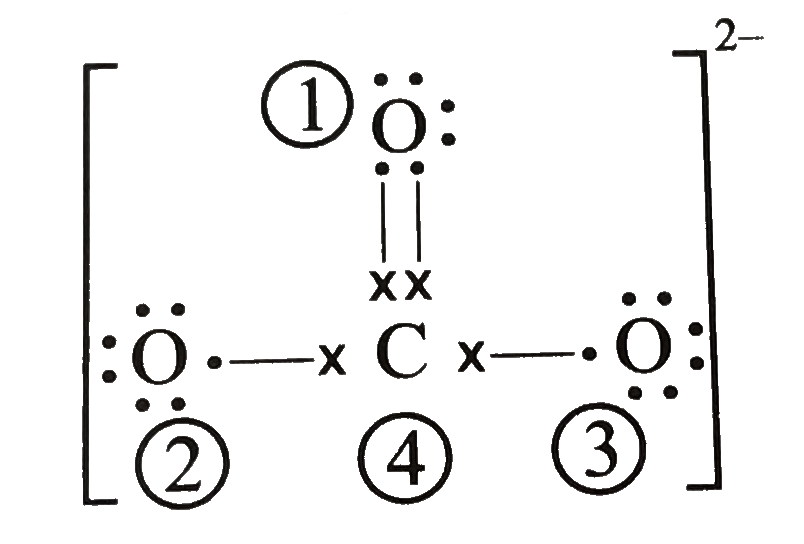
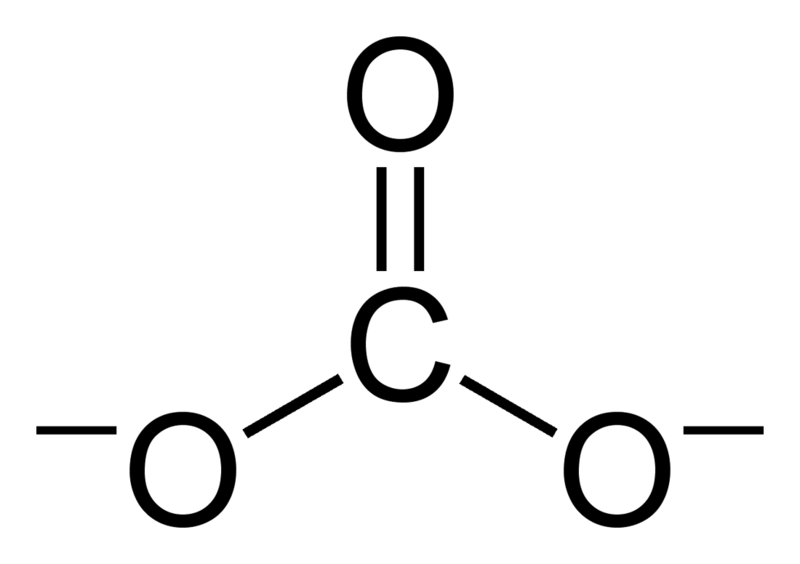
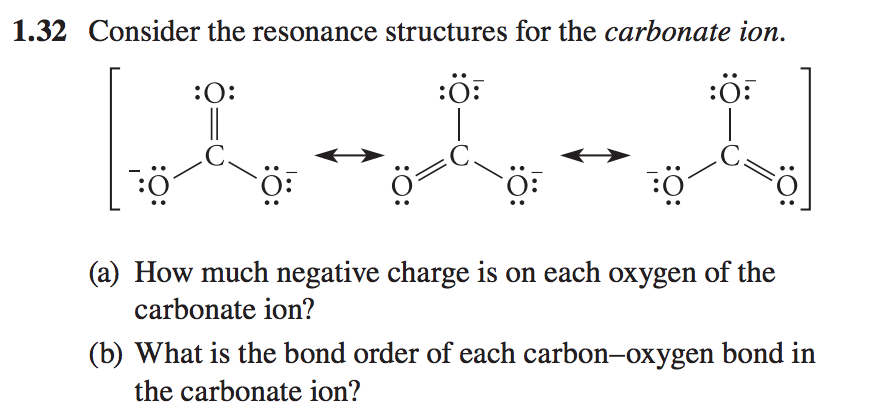
Determine the Lewis structure of CO32- (including formal charge charges on all atoms and resonance structures). | Homework.Study.com
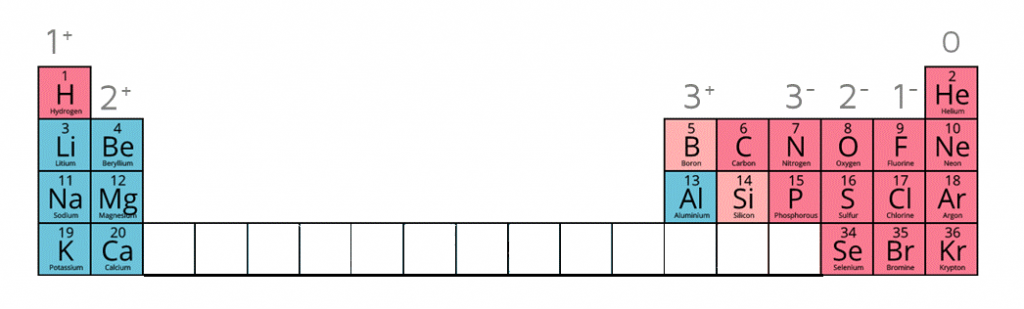
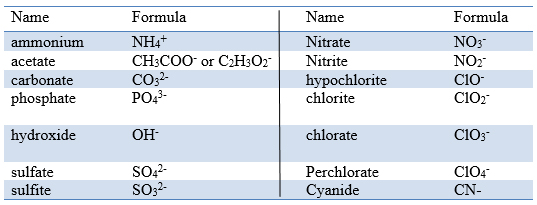
1:38 know the charges of these ions: metals in Groups 1, 2 and 3, non-metals in Groups 5, 6 and 7, Ag⁺, Cu²⁺, Fe²⁺, Fe³⁺, Pb²⁺, Zn²⁺, hydrogen (H⁺), hydroxide (OH⁻), ammonium (